MODELS OF THE ATOM
We all know that atoms are made of protons, neutrons and electrons? But how on earth do we know that if we can't see them? That's the amazing power of science! But it's a long story, with many false models along the way..... Join us as we explore the history of the atom over thousands of years...... Click on our interactive timeline on the relevent image to find out about particular chapters in the story! Watch the videos below for a quick TIKY TOCKY brain filler! However you like to learn...we have it here! All part of the service!


SCIENCE POETRY?
Science expressed through poetic prose! A new way to learn stuff through an ancient art. Click the link below.
SCIENCE SUMMARIES?
Try our videos. Made for the modern day student with a short attention span and lots to do!

MODELS OF THE ATOM! THE VIDEOS!

Ok you "Tiky Toky insta snap kids". Here are two short, yet epic videos explaining the history of the models of the atom in classic social media style to prevent loss of attention! Enjoy! If you are still reading this.....
DEMOCRITUS!


Democritus was basically the ancient Greek kid who wouldn’t stop breaking things to see what they were made of. He imagined taking, say, a piece of cheese (because Greeks loved cheese) and cutting it in half… then cutting that in half… and again… and again… until eventually you’d reach a teeny‑tiny bit you couldn’t cut anymore.
He called that final, uncuttable speck an atom — which literally means “cannot be cut.”
So while everyone else was busy inventing democracy and arguing about philosophy, Democritus was essentially doing the world’s first “What if I just keep chopping this?” science experiment. And from that slightly chaotic thought experiment, the idea of atoms was born.
After that no one actually talked much about atoms very much for a couple of thousand years.......
DALTON 1803
John Dalton was basically the organised friend who shows up after Democritus’ wild “let’s‑keep‑chopping‑stuff” idea and says, “Right, brilliant… but let’s make this scientific.”
He added three big upgrades:
Atoms aren’t just tiny mystery specks — they’re tiny balls with specific masses, like the world’s most microscopic bowling set.
All atoms of the same element are identical, so every oxygen atom is a perfect twin of every other oxygen atom.
Atoms join together in fixed ratios to make compounds, like strict Lego rules: two hydrogens plus one oxygen makes water, no freestyle building allowed.
Dalton basically turned Democritus’ thought experiment into a proper scientific model — less “Greek philosopher with a cheese knife,” more “19th‑century science teacher with a very neat notebook.




THE DISCOVERY OF THE ELECTRON -1897
JJ Thomson’s big breakthrough came from his experiments with cathode rays — mysterious greenish beams produced inside vacuum tubes. Scientists at the time weren’t even sure whether these rays were waves, particles, or just Victorian‑era weirdness.
Thomson investigated them carefully and showed that:
Cathode rays were actually tiny, negatively charged particles, not waves.
These particles were much smaller than atoms, meaning atoms weren’t indivisible after all.
He named these particles electrons, the first fundamental particles ever discovered.
Because electrons were negative, he reasoned that atoms must also contain positive charge to balance them.
This completely changed the atomic model: instead of solid spheres, atoms now had internal structure with sub‑atomic particles inside.




THE PLUM PUDDING MODEL!
Atoms aren’t solid spheres. Instead, Thomson said an atom is a ball of positive charge with tiny negative electrons dotted throughout it. The electrons are like little “raisins,” and the positive charge is like the soft “pudding” holding them in place.
So in this model:
The positive charge is spread out evenly through the whole atom.
The electrons sit inside it, spaced out but stuck there.
The atom is still neutral overall, because the positive and negative charges balance.
It was the first proper attempt to show internal structure inside the atom — even though later experiments proved it wasn’t quite right.




Here is a picture of Thomson's Plum Pudding Model and an actual plum pudding so you can see where it got its weird name from.....Write your text here...
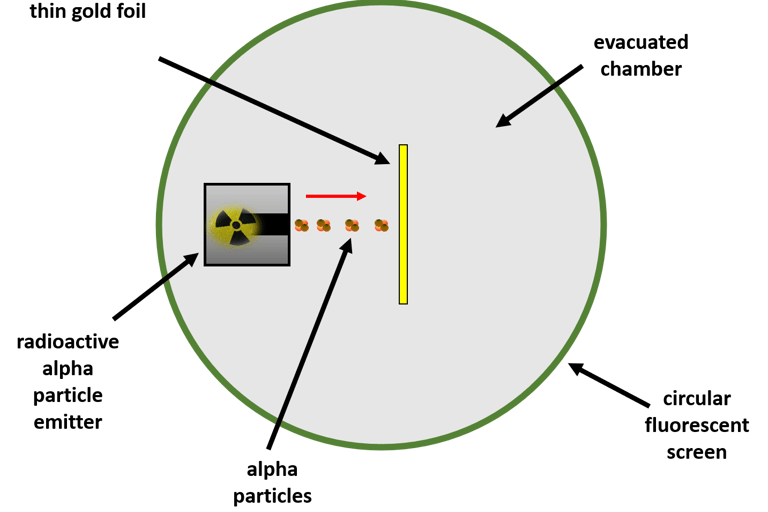
Rutherford’s Alpha‑Scattering Experiment
New Zealand Born Ernest Rutherford took one look at Thomson’s plum‑pudding model and essentially said, “Let’s test it properly.” What followed was one of the most famous experiments in science. His mates Geiger and Marsden also helped him.
Rutherford and his team fired alpha particles (tiny, fast, positively charged particles) at an extremely thin sheet of gold foil. They expected the particles to pass straight through with only tiny wobbles — exactly what the plum‑pudding model predicted.
What Rutherford observed
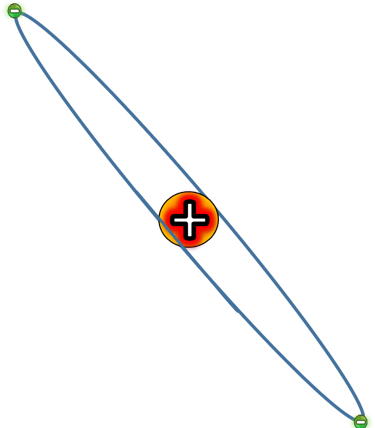
Most alpha particles went straight through the foil.
Some were deflected slightly.
A tiny number bounced straight back as if they’d hit something solid.
This was completely impossible under Thomson’s model.
What Rutherford concluded
Atoms are mostly empty space — that’s why most particles passed through.
There must be a small, dense, positively charged centre that repelled some particles.
This centre became known as the nucleus.
Almost all the atom’s mass is concentrated in this nucleus.
Rutherford’s work replaced the plum‑pudding model with the nuclear model — a huge leap forward in understanding atomic structure.
NOTE: HE DID NOT DISCOVER PROTONS OR NEUTRONS AT THIS STAGE!!!
SPOILER ALERT!!!



But the results were… surprising.........
Why not investigate what happened your self using this wonderful interactive masterpiece from our wonderful friends at Phet!
THE BOHR MODEL
Niels Bohr stepped in after Rutherford and solved the big problem with the nuclear model: if electrons were whizzing around the nucleus randomly, they should lose energy and spiral inwards. Atoms would collapse — which, awkwardly, they don’t.
Bohr’s Big Idea
Bohr proposed that electrons don’t orbit anywhere they like. Instead, they sit in fixed energy levels, or shells, around the nucleus.
What Bohr added
Electrons can only exist in specific shells, not in between them.
Each shell has a fixed amount of energy.
Electrons can jump between shells by absorbing or releasing energy (like climbing or dropping between floors in a building).
This explained why atoms give off specific colours of light — each jump produces a precise energy change.
Bohr’s model made the atom feel structured, stable, and predictable — a huge step forward from Rutherford’s “electrons somewhere out there” picture.






1. Democritus (around 400 BCE)
Suggested that matter is made of tiny, uncuttable particles called “atomos”
Used thought experiments rather than real experiments
Started the idea that atoms exist
2. John Dalton (1803)
Proposed the first scientific atomic theory
Said atoms are solid spheres with different masses
Showed that atoms combine in fixed ratios to form compounds
3. JJ Thomson (1897)
Investigated cathode rays and discovered the electron
Showed atoms contain tiny negative particles smaller than atoms
Proposed the plum pudding model with electrons in positive charge
4. Ernest Rutherford (1911)
Used the alpha‑scattering experiment on gold foil
Found that most of the atom is empty space
Discovered the nucleus: small, dense, and positively charged
5. Niels Bohr (1913)
Proposed electrons orbit in fixed energy levels (shells)
Explained why atoms give off specific colours of light
Made the nuclear model stable and predictable
6. Discovery of the Proton (1917)
Rutherford showed the nucleus contains positive particles called protons
Explained why the nucleus has a positive charge
Helped distinguish one element from another by proton number
7. Discovery of the Neutron (1932)
James Chadwick discovered the neutron
Showed the nucleus contains neutral particles as well as protons
Explained atomic mass and why some atoms are isotopes










